CLAYENS HEALTHCARE, representing the healthcare activity of the CLAYENS group, is the privileged partner of recognized healthcare actors.
Regardless of their industry (pharmaceutical companies, diagnostic laboratories, medical device manufacturers...) and their size (multinational or innovative start-up), we accompany our clients and ensure the development of their medical devices and pharmaceutical packaging.
Our recognized expertise in plastic injection and machining allows us to offer demanding clients comprehensive and tailor-made support in accordance with international regulatory requirements (ISO 13485- 2016, ISO 15378 and QSR - 21 CFR part 820).
We support our clients from the design of their device to its production: Design, prototyping, industrialization, qualification & validation, regulatory support, and production.
Among the 33 sites of the group, 12 have activity in healthcare and we have 9 cleanrooms in France (ISO7), Mexico (ISO7), Poland (ISO7) and the USA (ISO8).
Do you have a project?
CLAYENS Healthcare Services

Research & Development
Our multidisciplinary R&D team is capable of intervening at all stages of your project's development, from concept to market launch.
Industrialization
We industrialize our partners' projects by developing customized processes to transform the material, assemble and package the medical device.
Qualification, Validation & Regulatory Support
Our products are developed and manufactured in accordance with the requirements of international Health standards (ISO 13485, ISO 15378, FDA 21 CFR part 820).
Production
Our core business is the production of medical devices or technical components.
Our 12 manufacturing sites and 9 cleanrooms enable us to meet all of our clients' requirements.
Medical Device &/or Component
We develop primary and secondary pharmaceutical packaging, medical devices, and in vitro diagnostic devices, from their design to their production.
Our areas of expertise
We develop and manufacture products for...
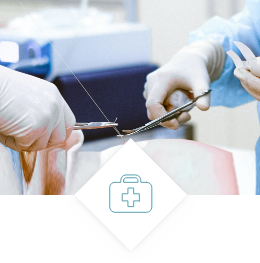
Medical devices
- Implant packaging
- Implants
- Hospital kits
- Implant injectors
- Surgical kits
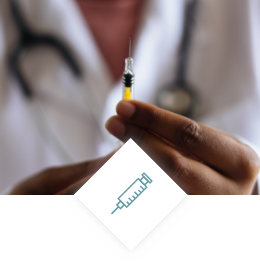
Pharmaceutical packaging
- Injectable drug delivery systems
- Injectables packaging & Accessories
- Respiratory drug delivery systems

CLAYENS business lines
Quality in the medical field is our first concern

Automotive

Industry & Equipment

Healthcare

Aerospace & Defense

Electrical & Infrastructure